Solution of Quadratic Equation by factorization
In this method, write the quadratic equation in the standard form as ax2 + bx + c = 0 (i) If two numbers r and s can be found for the equation (i) such that r + s = b and rs = ac then ax2 + bx + c can be factorized into two…

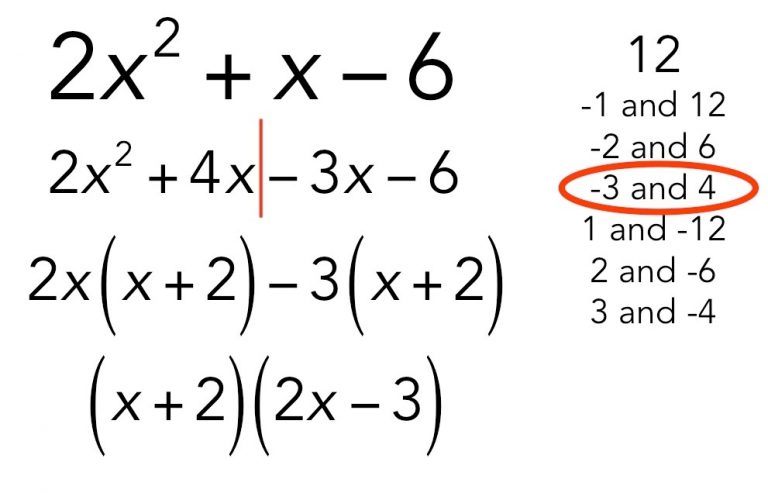


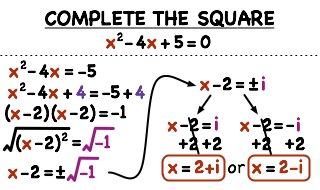




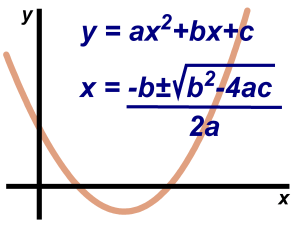
You must be logged in to post a comment.