Chemical Equilibrium
Generally, we presume that most chemical and physical changes proceed to completion. A complete reaction is one in which all reactants have been converted into products. However, most chemical reactions do not go to completion because products react themselves to form the reactants. As a result, after sometime no further change takes place. Quantities of reactants and products remain unchanged, and it seems that the reaction has stopped. In fact, these reactions do not stop; rather they take place on both directions at equal rate and attain the equilibrium state. Such reactions are called reversible reactions.
Many examples of physical and chemical equilibrium are found in nature.




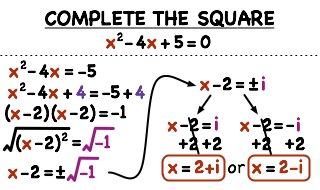


You must be logged in to post a comment.